 (timeline of theories from 2500 yrs ago to modern theory. The Starck Effect and Zeeman Effect was not explainable because the concept of sub energy levels was not introduced. Take a look at this image of hydrogen's probability clouds. If you look carefully you can also observe the standing wave type formation of the clouds. The Bohr Model is known as a planetary model because these orbits. In the Bohr model, the electrons travel in defined circular orbits around the small positively-charged nucleus. The probability clouds satisfy the Heisenberg uncertainty principle because the cloud only illustrate the probability of finding an electron in that space, not the certainty of finding it there. The Bohr model was also the first atomic model to incorporate quantum theory, meaning that it’s the predecessor of today’s more accurate quantum-mechanical models. Scientists now use the idea of a probability cloud to describe electron distribution around the nucleus. If the orbitals are circular, the 3D structure of an atom is not probable.Īn application of Schrödinger's equation to hydrogen's emission / absorption spectra it was shown that a standing wave energy level model corresponded to the emitted / absorbed photons. 
And also, it does not satisfy the fact that atoms have a 3D formation. This does not satisfy the Heisenberg uncertainty principle, which is that it is not possible to know the position and momentum of a particle simultaneously. 
The main problem lies in the idea of electrons in circular orbits. In principle the quantification aspect of the model is still believed to be correct. (b) Atoms with mass number but different atomic numbers are called isobars. Atoms with same mass number but different atomic numbers are called. (b) Number of p number of e 89 and neutrons 231 89 142. He believed that electrons moved around the nucleus in circular orbits with quantised potential and kinetic energies. Number of protons, neutrons and electrons in the element 89 X 231 is. Bohr's most significant contribution was explaining the model using the quantification energy. In the ground state, the electron goes round the proton in a circle of radius 5.3 × 10 11 m. The centripetal force is provided by the Coulomb attraction. The proton itself is assumed to be fixed in an inertial frame. 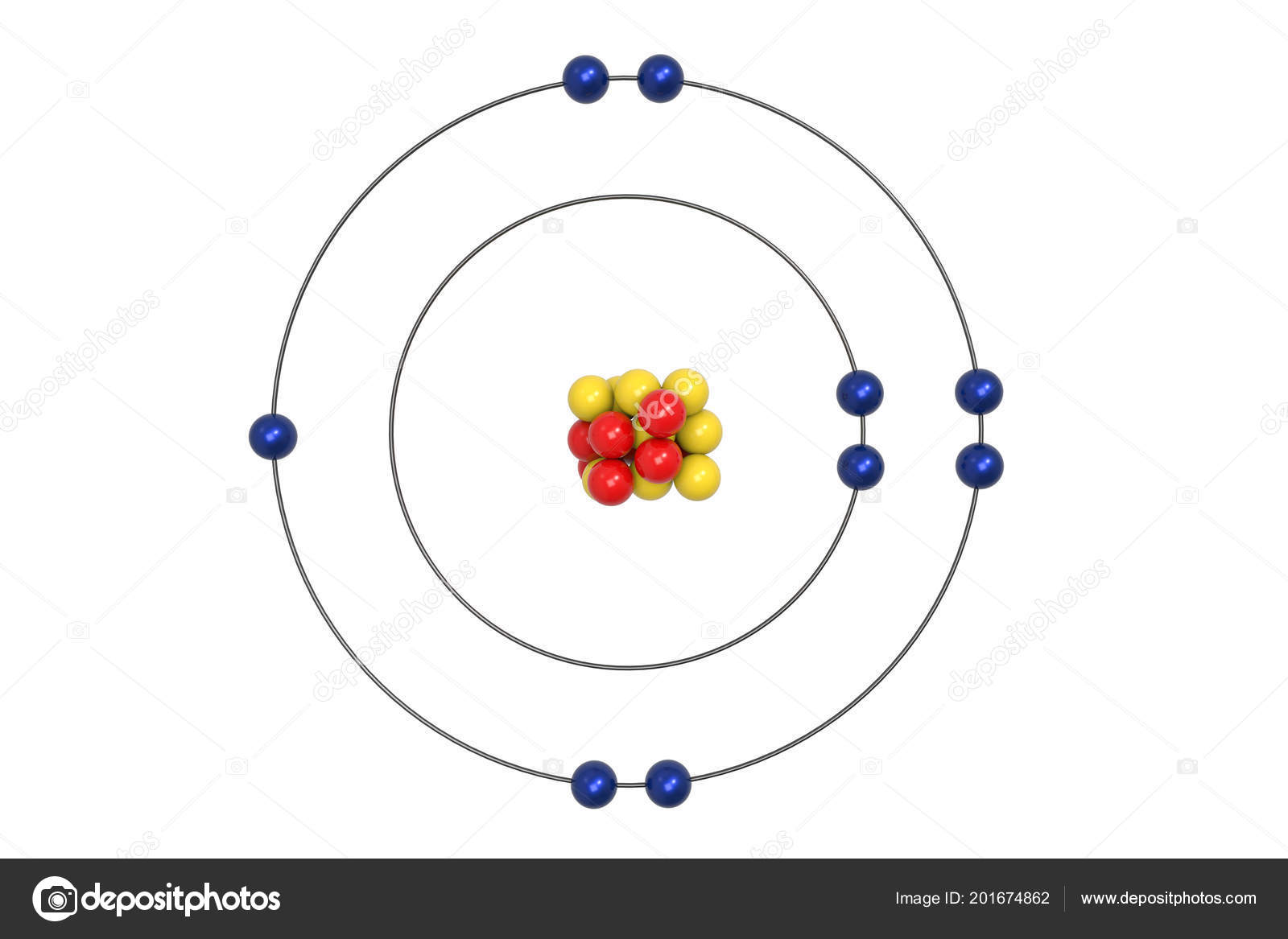
In it most of the atom's mass is concentrated into the center (what we now call the nucleus ) and electrons surround the positive mass in something like a cloud. In the Bohr model of hydrogen atom, the electron is treated as a particle going in a circle with the centre at the proton. It would be like a ladder that had rungs only at certain heights.Bohr thought that electrons orbited the nucleus in circular paths whereas in the modern view atomic electron structure is more like 3D standing waves.īohr built upon Rutherford's model of the atom. Electrons in a quantized orbit will not radiate energy. The nucleus is of infinite mass and is rest. 
The electron in a hydrogen atom travels around the nucleus in a circular orbit. The Bohr model was based on the following assumptions. The energy levels are quantized, meaning that only specific amounts are possible. In 1913, Niels Bohr proposed a model for the hydrogen atom. Note that the spacing between rungs gets smaller at higher energies (CC BY-NC Ümit Kaya)īohr hypothesized that the only way electrons could gain or lose energy would be to move from one energy level to another, thus gaining or losing precise amounts of energy. \): The energy levels of the electrons can be viewed as rungs on a ladder. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
